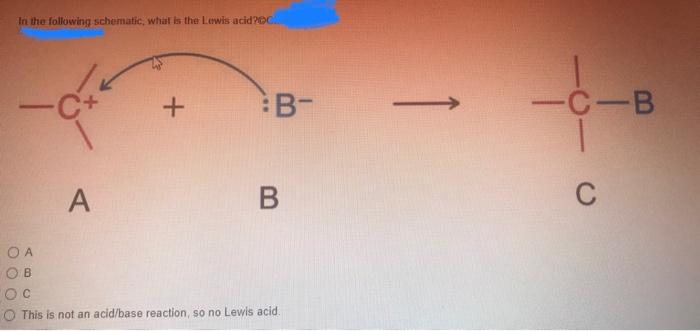
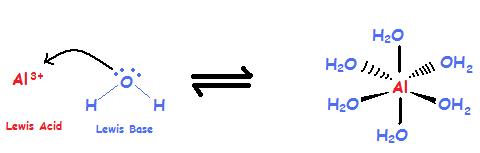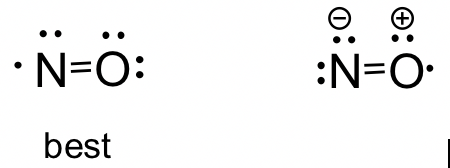Lewis acid-assisted reduction of nitrite to nitric and nitrous oxides via the elusive nitrite radical dianion | Nature Chemistry

Ni‐Ti Intercalated and Supported Bentonite for Selective Hydrogenation of Cinnamaldehyde - Xu - ChemPhysChem - Wiley Online Library
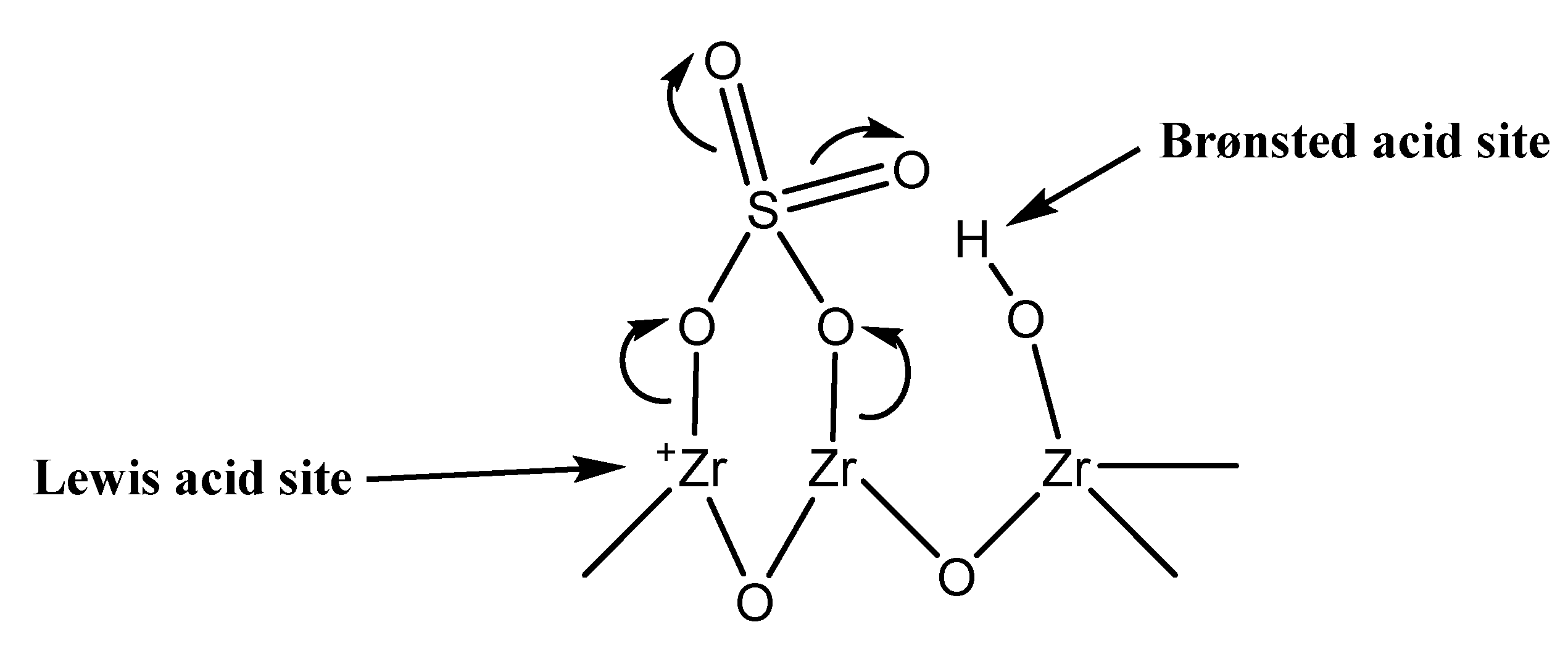
Nanomaterials | Free Full-Text | Comparative Study of Different Acidic Surface Structures in Solid Catalysts Applied for the Isobutene Dimerization Reaction

BJOC - Post-synthesis from Lewis acid–base interaction: an alternative way to generate light and harvest triplet excitons

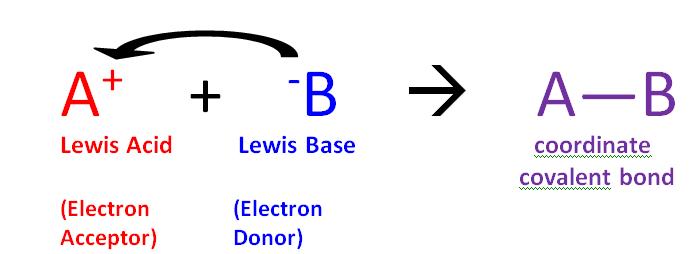
SOLVED: Question 12 Not vet answered Identify the Lewis Acid and Base in the reaction of Zn2+ and CN" to form Zn(CN)2: Points out of 5 Select one: a.Zn(CN)z is the Lewis

Role of Brønsted and Lewis acid sites on Ni/TiO2 catalyst for vapour phase hydrogenation of levulinic acid: Kinetic and mechanistic study - ScienceDirect

Which of these two compounds, trimethyl boron, and triphenyl boron is a stronger Lewis acid when reacted with ammonia? | Homework.Study.com

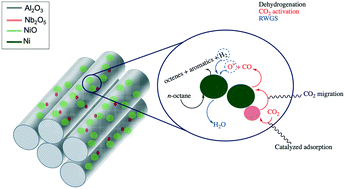
Combining Ni3P and Lewis Acid–Base Pair as a High-Performance Catalyst for Amination of 1-Octanol | SpringerLink

Design of Lewis Acid Centers in Bundlelike Boron Nitride for Boosting Adsorptive Desulfurization Performance | Industrial & Engineering Chemistry Research