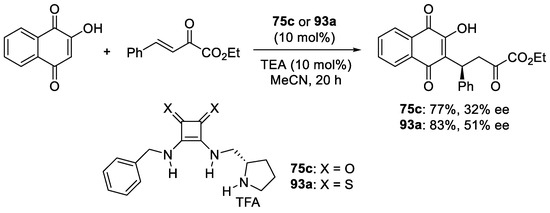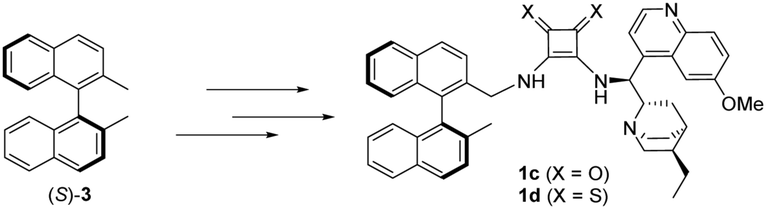
New enantiopure binaphthyl-cinchona thiosquaramides: synthesis and application for enantioselective organocatalysis - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ06451B
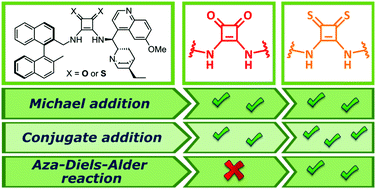
New enantiopure binaphthyl-cinchona thiosquaramides: synthesis and application for enantioselective organocatalysis - New Journal of Chemistry (RSC Publishing)

Chiral Bifunctional Thiosquaramides as Organocatalysts in the Synthesis of Enantioenriched 3,3‐Disubstituted Oxindoles - Rodríguez‐Ferrer - 2019 - European Journal of Organic Chemistry - Wiley Online Library

Thiosquaramide-Based Supramolecular Polymers: Aromaticity Gain in a Switched Mode of Self-Assembly | Journal of the American Chemical Society

Thiosquaramide-Based Supramolecular Polymers: Aromaticity Gain in a Switched Mode of Self-Assembly | Journal of the American Chemical Society

Molecules | Free Full-Text | Recent Advances in Asymmetric Synthesis of Pyrrolidine-Based Organocatalysts and Their Application: A 15-Year Update

Development of Chiral, Bifunctional Thiosquaramides: Enantioselective Michael Additions of Barbituric Acids to Nitroalkenes | Journal of the American Chemical Society

Dicyclopentyl Dithiosquarate as an Intermediate for the Synthesis of Thiosquaramides | Organic Letters

Development of Chiral, Bifunctional Thiosquaramides: Enantioselective Michael Additions of Barbituric Acids to Nitroalkenes | Journal of the American Chemical Society

Thiosquaramide-Based Supramolecular Polymers: Aromaticity Gain in a Switched Mode of Self-Assembly | Journal of the American Chemical Society
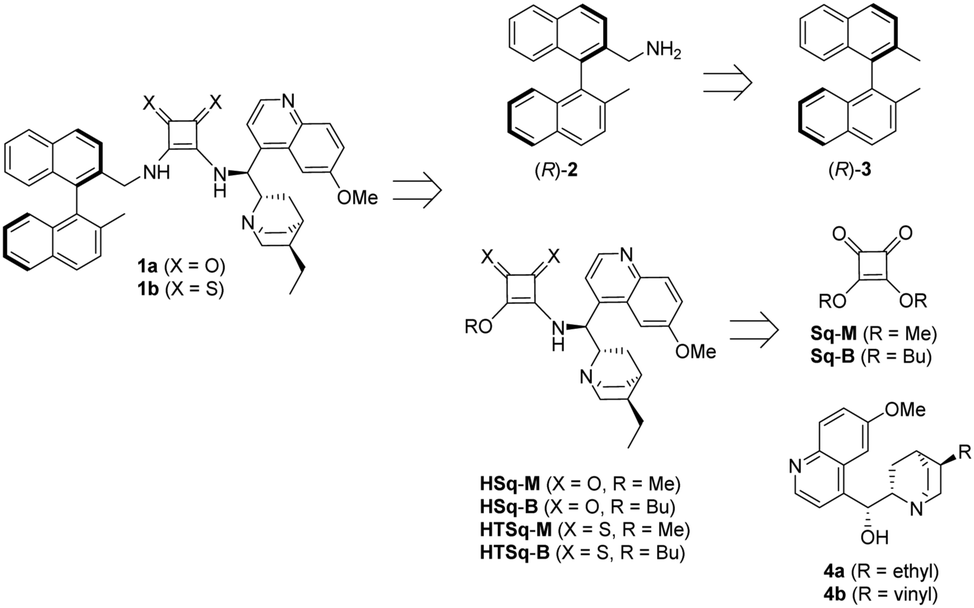
New enantiopure binaphthyl-cinchona thiosquaramides: synthesis and application for enantioselective organocatalysis - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ06451B

Optimised geometries and schematic structures of binaphthyl-cinchona... | Download Scientific Diagram

Dicyclopentyl Dithiosquarate as an Intermediate for the Synthesis of Thiosquaramides | Organic Letters

Dicyclopentyl Dithiosquarate as an Intermediate for the Synthesis of Thiosquaramides | Organic Letters
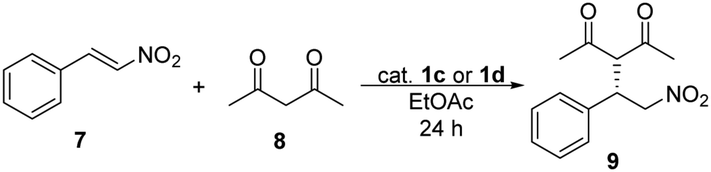
New enantiopure binaphthyl-cinchona thiosquaramides: synthesis and application for enantioselective organocatalysis - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ06451B

Thiosquaramide-Based Supramolecular Polymers: Aromaticity Gain in a Switched Mode of Self-Assembly | Journal of the American Chemical Society
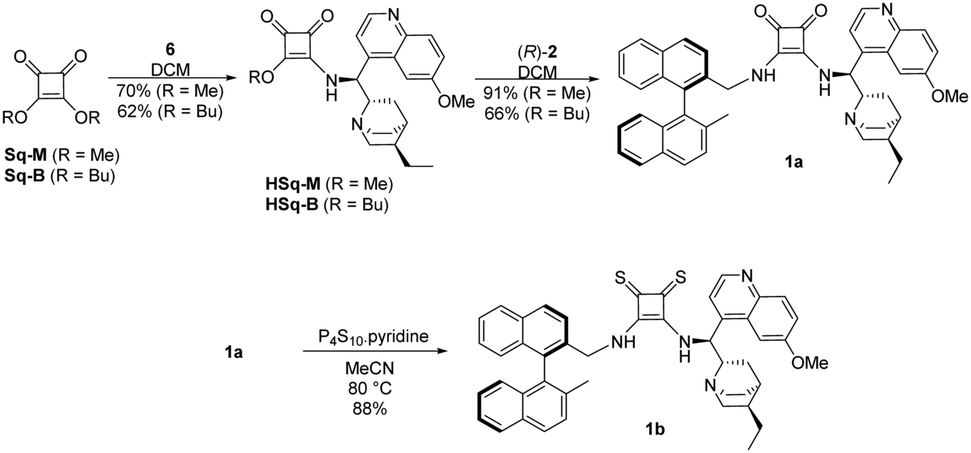
New enantiopure binaphthyl-cinchona thiosquaramides: synthesis and application for enantioselective organocatalysis - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ06451B

PDF) Comparison of Cinchona Catalysts Containing Ethyl or Vinyl or Ethynyl Group at Their Quinuclidine Ring

Development of Chiral, Bifunctional Thiosquaramides: Enantioselective Michael Additions of Barbituric Acids to Nitroalkenes | Journal of the American Chemical Society